Christopher Lu Lab
- Acute kidney injury
- Renal transplant rejection
- Inflammation in chronic kidney disease
Ischemic acute kidney injury, or AKI (previously called "acute renal failure" or "acute tubular necrosis") affects approximately 25 percent of ICU patients. AKI confronts the physician with major clinical problems, and the scientist with fundamental unsolved questions. The physician is confronted by a disease with no accepted treatment other than supportive care such as dialysis. The acute mortality and morbidity remains high, and the long-term problems include progressive chronic kidney disease and long-term mortality. The scientist is confronted by a disease where the underlying pathophysiology is not understood. Although it is now recognized that the initial insult to the kidney is exacerbated by a maladaptive inflammatory response, how injury is translated into inflammation remains a fundamental unsolved problem.
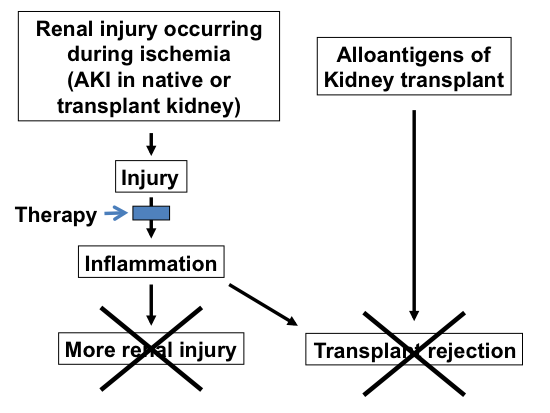
Furthermore, ischemic AKI inevitably occurs during the process of renal transplantation - donor hypotension, cold storage, and transplant surgery. The inflammatory response to this allograft ischemia will include recipient leukocytes that should exacerbate allograft rejection. Thus, our studies will also help us understand and treat allograft rejection.
Ischemic kidney injury occurs both in the native, non-transplanted kidney and during renal transplantation (organ procurement, storage, and recipient surgery). Understanding how ischemic injury leads inflammation may lead to therapies that ameliorate ischemic AKI in the intensive care unit and also ameliorate transplant rejection. Note that transplant rejection requires two signals – alloantigens plus inflammation caused by injury during the transplant process.
The overall goal of our laboratory is to elucidate this unsolved problem in the hope that this will led to more effective therapies. To this end, we use in vivo models of ischemic AKI in mice, and in vitro model systems to perform detailed studies of proinflammatory genes activated by renal ischemia/ reperfusion. We have found that molecules, normally residing within healthy cells, are released into the extracellular space when renal cells are injured by ischemia in vivo and in vitro. These molecules activate proinflammatory genes. In addition, we study the effects of reactive oxygen species, produced during ischemia-reperfusion, on proinflammatory genes.
We are also translating this bench research to the bedside by studying pre-transplant renal biopsies; these biopsy studies will determine if the genes associated with the inflammatory response to ischemia in mice also are activated in humans. In addition to addressing fundamental questions, these clinical studies may predict the course and guide therapy of renal transplant recipients.
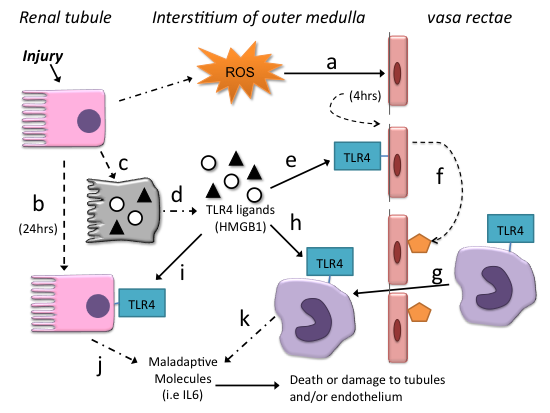
TLR4-HMGB1 in Ischemic AKI. In response to reactive oxygen species (ROS) released during ischemia/ reperfusion, endothelia of the vasa rectae express TLR4 within 4 hours after reperfusion (a). Renal tubules also express TLR4, but only after 24 hours following reperfusion; renal tubular TLR4 expression is a response to interferon gamma and TNFα (b). Injury also increases renal tubular production of endogenous TLR4 ligands such as HMGB1 (b), and severely injured cells release these ligands into the extracellular space (d). These extracellular TLR4-ligands trigger maladaptive responses. They activate TLR4 on endothelial cells (e) which in turn, express adhesion molecules (f) that facilitate diapedesis of monocytes (macrophages) from blood into the renal interstitial space (g). The endogenous TLR4-ligands (HMGB1) then activate TLR4 on macrophages (h), and tubules (i). The activated macrophages and tubules release maladaptive molecules such as interleukin 6 (j & k) that exacerbate injury.

Department of Internal Medicine
UT Southwestern Medical Center
5323 Harry Hines Blvd.
Dallas, TX 75390
C. Y. Lu, P. D. Winterberg, J. Chen and J. R. Hartono. Acute kidney injury: a conspiracy of toll-like receptor 4 on endothelia, leukocytes, and tubules. Pediatr Nephrol. 2012;27:1847-54.
C. Y. Lu, J. Hartono, M. Senitko and J. Chen. The inflammatory response to ischemic acute kidney injury: a result of the 'right stuff' in the 'wrong place'? Curr Opin Nephrol Hypertens 2007;16(2):83-9. Epub 2007/02/13.
C. Y. Lu, J. G. Penfield, M. L. Kielar, M. A. Vazquez and D. R. Jeyarajah. Perspectives in basic science: Hypothesis: is renal allograft rejection initiated by the response to injury sustained during the transplant process? Kidney Int 1999;55:2157-2168.
R. W. Redline and C. Y. Lu. Role of local immunosuppression in murine fetoplacental listeriosis. J Clin Invest 1987;79:1234-1241.
C. Y. Lu, E. G. Calamai and E. R. Unanue. A defect in the antigen-presenting function of macrophages from neonatal mice. Nature 1979;282:327-329.
P. D. Winterberg, Y. Wang, K. M. Lin, J. R. Hartono, G. T. Nagami, X. J. Zhou, J. M. Shelton, J. A. Richardson and C. Y. Lu. Reactive-oxygen species (ROS) and IRF1 stimulate IFNalpha production by proximal tubules during ischemic AKI. Am J Physiol Renal Physiol. 2013;305(2):F162-72.
J. Chen, M. M. Matzuk, X. J. Zhou and C. Y. Lu. Endothelial pentraxin 3 contributes to murine ischemic acute kidney injury. Kidney Int. 2012; 82:1195-1207.
J. Chen, R. John, J. A. Richardson, J. M. Shelton, X. J. Zhou, Y. Wang, Q. Q. Wu, J. R. Hartono, P. D. Winterberg and C. Y. Lu. Toll-like receptor 4 regulates early endothelial activation during ischemic acute kidney injury. Kidney Int. 2011; 79(3):288-99.
J. Chen, J. Hartono, R. John, M. Bennett, X. Zhou, Y. Wang, Q. Wu, P. Winterberg, G. T. Nagami and C. Y. Lu. Interleukin 6 production by leukocytes during ischemic acute kidney injury is regulated by TLR4. Kidney Int. 2011; 80(5):504-15.
Q. Q. Wu, Y. Wang, M. Senitko, C. Meyer, W. C. Wigley, D. A. Ferguson, E. Grossman, J. Chen, X. J. Zhou, J. Hartono, P. Winterberg, B. Chen, A. Agarwal and C. Y. Lu. Bardoxolone methyl (BARD) ameliorates ischemic AKI and increases expression of protective genes Nrf2, PPARgamma, and HO-1. Am J Physiol Renal Physiol. 2011; 300(5):F1180-92.
Y. Wang, R. John, J. Chen, J. A. Richardson, J. M. Shelton, M. Bennett, X. J. Zhou, G. T. Nagami, Y. Zhang, Q. Q. Wu and C. Y. Lu. IRF-1 promotes inflammation early after ischemic acute kidney injury. J Am Soc Nephrol 2009; 20(7):1544-55.