Research in the Mendelson Lab focuses on molecular mechanisms involved in developmental and hormonal regulation of key genes and signaling pathways in perinatal biology and female reproduction. We study genetic and epigenetic mechanisms in differentiation of the surfactant-producing cells of fetal lung and placental syncytiotrophoblast, as well as mechanisms underlying the maintenance of myometrial quiescence during pregnancy and the initiation of uterine contractility leading to term and preterm labor. Our studies support an important role of maturation of the fetal lung and synthesis of lung surfactant lipids and proteins in the timing of birth.
Developmental induction of type II cell differentiation and surfactant lipoprotein synthesis by the fetal lung are essential for newborn survival. In addition to their vital role to reduce alveolar surface tension after birth, surfactant components protect the alveolar epithelium from the inflammatory consequences of exposure to high O2 tension and inhaled pathogens. We observed that the major surfactant protein, SP-A (encoded by the SFTPA gene), an immune modulator, is developmentally upregulated in fetal lung with type II cell differentiation and surfactant glycerophospholipid synthesis. SP-A expression and type II cell differentiation in cultured human fetal lung (HFL) epithelial cells are stimulated by cAMP and inhibited by transforming growth factor TGF-β and hypoxia. In studies using transgenic mice and transfected type II cells, we identified a conserved ~300-bp region upstream of the rabbit and human SFTPA genes that is critical for lung cell-specific, developmental and cAMP induced expression. This region serves as an ‘enhanceosome’ through which developmental and cAMP induction of hSFTPA promoter activity are mediated by cooperative interactions of transcription factors bound to key response elements (Fig. 1). The TBE (red) binds thyroid transcription factor 1 (TTF-1/Nkx2.1) and nuclear factor (NF)-κB in a cAMP-responsive manner, resulting in enhanced recruitment of the histone acetylase, CREB-binding protein (CBP), and steroid receptor coactivator (SRC)-1, with increased acetylation of histone H3K9. cAMP also enhances estrogen-related receptor ∝(ERRα) binding and transcriptional activity by increasing its interaction with protein kinase-A and SRC-2. In fetal mice doubly-deficient in Src-1 and Src-2, SP-A expression was significantly decreased. Whereas, O2-dependent cAMP induction of SP-A in HFL type II cells is mediated, in part, by TTF-1 and NF-κB binding to the SFTPA promoter, mechanisms for O2 regulation of type II cell differentiation and SFTPA expression are not fully understood. We recently observed that the redox-sensitive transcription factor, nuclear factor erythroid 2-related factor 2 (NRF2) and its co-regulated transcription factors, C/EBPβ and PPARγ, were markedly induced by cAMP in HFL type II cells in an O2-dependent manner. Moreover, NRF2 binds to an ‘anti-oxidant response element’ (ARE) in the hSFTPA promoter in cultured HFL epithelial cells (Fig. 1). In mouse fetal lung, a developmental increase in Nrf2, C/ebpβ and Pparγ, and a decrease in Nrf2 inhibitor Keap1 were observed between 14.5 and 19.5 (term) days post-coitum (dpc), with temporal induction of SP-A.
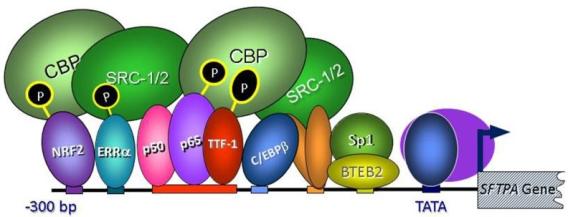 Key response elements in the SFTPA promoter comprise an enhanceosome that mediates tissue/cell-specific, developmental and hormonal regulation expression.
Key response elements in the SFTPA promoter comprise an enhanceosome that mediates tissue/cell-specific, developmental and hormonal regulation expression. As a component of the innate immune system, SP-A binds to and activates alveolar macrophages (MΦ) to facilitate pathogen clearance within the lung alveoli. SP-A also is an immune modulator that inhibits dendritic cell maturation and stimulates proliferation of immunosuppressive regulatory T cells (Treg, CD4+CD25+ FoxP3+). The immune functions of SP-A likely protect the postnatal lung from the inflammatory challenges imposed by exposure to reactive oxygen species (ROS) and microbial pathogens. In our ongoing research, we are testing the hypothesis that transcription factors, including NRF2, C/EBPβ, and PPARγ serve crucial roles in the developing fetal lung to promote type II cell differentiation and enhance production of SP-A and other key immune modulators that act on immune cells to protect the alveolar epithelium from oxidative and inflammatory stress. We postulate that decreased production of immune modulators by lungs of premature infants increase hyperoxic and inflammatory stress and enhance susceptibility to bronchopulmonary dysplasia (BPD) and other chronic lung diseases.
SP-A also can have proinflammatory actions. As described below, we have compelling evidence that secretion of surfactant components SP-A and the proinflammatory glycerophospholipid, platelet-activating factor (PAF), by the developing fetal lung near term, serve as fetal signals for the initiation of labor.
The multinucleated syncytiotrophoblast (SynT) of the human placenta is formed by fusion of underlying proliferating cytotrophoblasts (CytT). The SynT, which covers the chorionic villi, is bathed in maternal blood and performs several essential functions to ensure growth and survival of the developing embryo. These include transport of O2 and nutrients and synthesis and secretion of protein and steroid hormones, including estrogen and progesterone. Synthesis of estrogens from C19-steroids is catalyzed by aromatase P450 (product of the hCYP19A1 gene). The ability of the human placenta to synthesize estrogens is vastly increased after the ninth week of gestation, in association with CytT invasion and enlargement of the uterine arterioles, increased blood flow and O2 availability to the floating chorionic villi. The exceptionally high levels of placental aromatase likely function to metabolize large amounts of C19-steroids produced by the human fetal adrenals (e.g. dehydroepiandrosterone), thus preventing conversion of these steroids to active androgens, which can masculinize the fetus. Biologically active estrogens and their metabolites formed by placental aromatase may also enhance angiogenesis and uteroplacental blood flow and reduce systemic vascular resistance.
We have explored mechanisms that underlie human placental trophoblast differentiation and effects of O2 tension using the placenta-specific (PS) hCYP19A1/ aromatase promoter as a model. Trophoblast stem cells (TSC) and CytT do not express aromatase; however, when CytT fuse to form SynT, aromatase is markedly induced. By contrast, when CytT are cultured in a hypoxic (2% O2) environment, SynT differentiation and induction of hCYP19A1PS are prevented by increased binding of transcriptional repressors, MASH-2/ASCL2 (15) and USF1/2 (Fig. 2); these factors are degraded upon SynT differentiation in 20% O2. This likely allows assembly of an enhanceosome (Fig. 2) comprised of transcription factors Sp1, estrogen receptor α (ERα), estrogen-related receptor γ (ERRγ) and glial cells missing 1 (GCM1), which serves a crucial role in trophoblast development and SynT fusion. This complex also contains NRF2 (Fig. 2), which we found to be essential for O2-mediated aromatase mRNA induction in cultured trophoblasts.
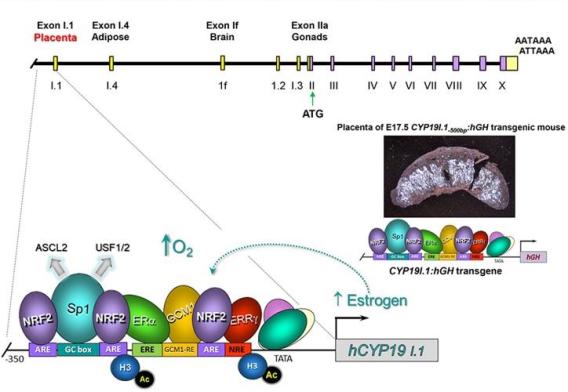 Fig. 2: Placenta-specific (PS) expression of hCYP19A1 aromatase is controlled by an enhanceosome immediately upstream of PS exon I.1 and 100 kb upstream of the translation start site in exon II.
Fig. 2: Placenta-specific (PS) expression of hCYP19A1 aromatase is controlled by an enhanceosome immediately upstream of PS exon I.1 and 100 kb upstream of the translation start site in exon II. The SynT also may sustain pregnancy through production of immune modulators, which promote immune tolerance at the maternal-fetal interface (MFI)/decidua, to protect the hemi-allogeneic fetus from rejection by the maternal immune system. However, the underlying mechanisms have not been fully defined. Using human trophoblasts in primary culture, we observed that several genes involved in the induction and maintenance of immune tolerance were markedly upregulated during differentiation of CytT to SynT. These include: heme oxygenase I (HMOX1); programmed death-ligand 1 (PD-L1); kynurenine pathway components, indoleamine 2,3-dioxygenase (IDO1) and arylhydrocarbon receptor (AhR). Intriguingly, we discovered that the O2-regulated transcription factors, NRF2, C/EBPβ, and PPARγ, were markedly induced when CytT fuse to form SynT. NRF2 knockdown blocked the induction of aromatase, as well as upregulation of C/EBPβ, PPARγ, and the above-mentioned immune modulatory factors (Fig. 3). NRF2 deficiency has been implicated in preeclampsia (PE), a hypertensive disorder of pregnancy associated with shallow implantation, placental hypoxia and inflammation. Mice with a global KO of Nrf2 were reported to manifest increased placental inflammatory factors, oxidative stress and susceptibility to preterm birth induction. Importantly, we recently found that placentas of Nrf2 KO mice at 12.5 dpc express decreased Pparγ, Hmox1, and Ahr mRNA.
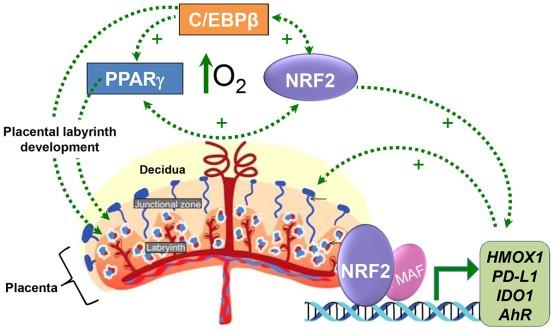 Fig. 3: NRF2, PPary and C/EBP act in an 02 mediated positive feedback loop to augment expression of immune modulators, which act at the MFI/dcidua to maintain tolerance to the hemi-allogeneic fetus
Fig. 3: NRF2, PPary and C/EBP act in an 02 mediated positive feedback loop to augment expression of immune modulators, which act at the MFI/dcidua to maintain tolerance to the hemi-allogeneic fetus Based on these and other findings we are utilizing human trophoblasts in primary culture, human trophoblast stem cells and gene-targeted mice to test the hypothesis that the redox-regulated transcription factor, NRF2, together with C/EBPβ and PPARγ, serve as ‘key regulators’ of the profound biochemical, genetic and epigenetic changes that underlie production of immune modulators. These modulators act on immune cells within the decidua to maintain an anti-inflammatory milieu and protect the fetus from rejection by the maternal immune system to maintain pregnancy. We further propose that increased inflammation at term or preterm causes decreased placental expression of immune modulators and a ‘break’ in tolerance leading to term or preterm labor or PE.
Preterm birth (6 births globally each year, is a major cause of death within the first month of postnatal life. The highest rates of preterm birth (≥15% of all live births) occur in sub-Saharan Africa, Pakistan and Indonesia. In the U.S., the preterm birth rate remains at ~10% of all live births, overall. However, significant racial disparities in preterm birth rates exist, with the incidence of preterm birth among African-Americans being 50% higher than that of the overall population. Notably, the underlying causes for these racial differences remain unknown. Astonishingly, the modalities used to treat and/or prevent preterm labor have changed little over the past 50 years. This is due, in part, to our incomplete understanding of mechanisms that mediate myometrial quiescence and contractility and the reluctance of pharmaceutical companies to engage in drug discovery in this critical area.
Our research is focused on the mechanisms for maintenance of myometrial quiescence during pregnancy and for the increase in uterine contractility leading to labor. We have observed that myometrial quiescence is controlled primarily by the anti-inflammatory and anti-contractile actions of progesterone (P4) (secreted by the placenta and/or the ovarian corpus luteum, depending upon the species), acting via its nuclear receptor (PR) (Fig. 4). As shown in the upper left, we found that P4/PR exerts anti-inflammatory effects by tethering to NF-κB p65 bound to NF-κB response elements (NF-κBRE) within the promoters of proinflammatory and contractile (CAP) genes (e.g. oxytocin receptor [OXTR], connexin 43 [CX43], cyclooxygenase 2 [COX2]). This stimulates recruitment of corepressors, such as GATAD2B. Similar mechanisms are believed to occur at AP-1 sites upstream of CAP gene promoters, whereby PR interacts with FOS/JUN and recruits corepressors. As shown in the upper right, P4/PR also inhibits NF-κB activation and proinflammatory and contractile gene expression in myometrium by binding to the promoter of the NF-κB inhibitor, IκBα, and enhancing its expression. P4/PR also exerts anti-inflammatory effects in myometrium by binding to the MKP1/DUSP1 promoter to increase its expression and prevent MAPK activation, resulting in the inhibition of NF-κB and AP-1 activity. As shown in the lower panel, P4/PR blocks contractile gene expression by binding to the promoter of the transcriptional repressor, ZEB1, and enhancing its expression. The increased levels of ZEB1 bind to the promoters of contractile genes to inhibit their expression, and activates expression of the microRNA, miR-199a/-214, cluster to inhibit expression of their target, COX-2. ZEB1 also inhibits miR-200 expression, leading to further induction of ZEB1 and ZEB2, as well as STAT5b, causing suppression of 20α-HSD, leading to increased local P4 accumulation.
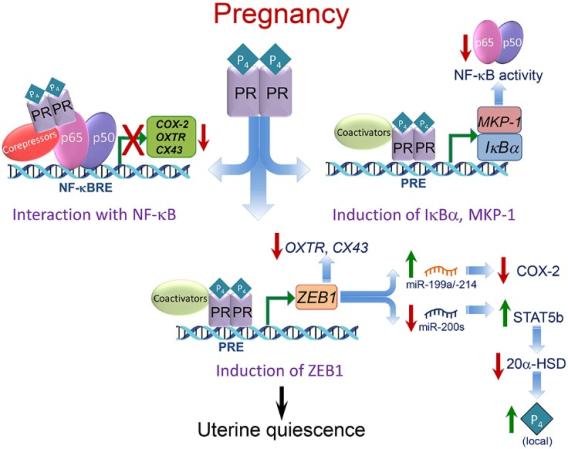 Fig. 4: Mechanisms whereby P4/PR maintains of myometrial quiescence during pregnancy.
Fig. 4: Mechanisms whereby P4/PR maintains of myometrial quiescence during pregnancy. Parturition in all species is associated with an increased inflammatory response and a decline in PR function within the myometrium. Near term, signals from the fetus (e.g. increased surfactant lipoprotein secretion) and mother (e.g. uterine stretch with increased chemokine production, increased estrogen receptor signaling), cause increased immune cell invasion of the fetal membranes, decidua, and myometrium. At preterm, immune cell invasion is stimulated by ascending infection associated with chorioamnionitis, as well as increased uterine stretch in twin or multi-fetal pregnancies. The invading immune cells produce proinflammatory cytokines that cause activation of inflammatory transcription factors, such as NF-κB and AP-1, which promote activation of proinflammatory and CAP gene expression and a decline in PR function, caused by a decrease in coactivators and increased expression of truncated PR isoforms. The decline in PR function results in decreased ZEB1 expression, with increased expression of CAP genes and miR-200 family members and decreased expression of the miR-199a/214 cluster. The increase in miR-200s suppress their targets ZEB1/2 and STAT5b and increase local P4 metabolism via upregulation of 20α-HSD. The decrease in miR-199a/214 results in upregulation of their target, COX-2, and an increase in synthesis of contractile prostaglandins. These events culminate in an increase in myometrial contractility leading to parturition.
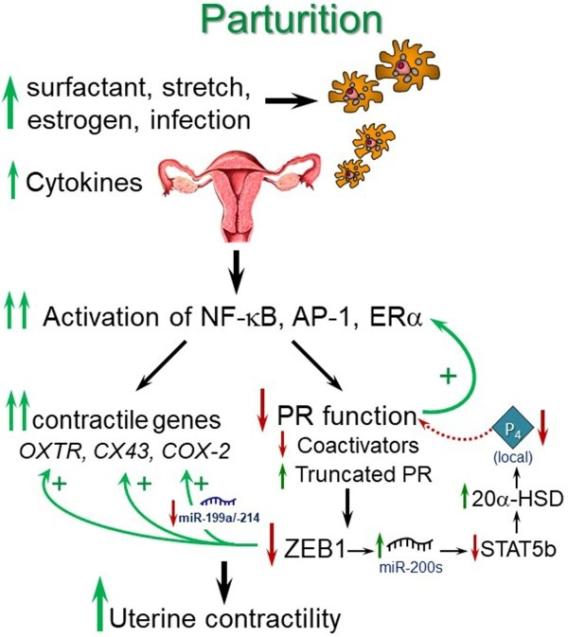 Fig. 5: Labor at term or preterm is associated with an inflammatory response.
Fig. 5: Labor at term or preterm is associated with an inflammatory response. Studies are ongoing to define the molecular mechanisms whereby key transcriptional coregulators mediate hormonal regulation of myometrial quiescence and contractility, and to identify and characterize novel genes that control myometrial quiescence and contractility.
Signals for the initiation of parturition at term have long remained a mystery. Our work suggests that the fetus signals the mother when it is ready to be born through production of surfactant components by the maturing lung. We observed that the major surfactant protein, SP-A, secreted in increasing amounts by the mouse fetal lung into amniotic fluid (AF) after 17.5 days post-coitum (dpc) (term = 19.5 dpc), serves as a hormonal signal for parturition. Our prior studies revealed that the temporal induction of SP-A secretion was associated with increased proinflammatory cytokine production by amniotic fluid (AF) MΦ, myometrial NF-κB activation and increased contractile gene expression. Accordingly, mice carrying a targeted deletion of the SP-A (Sftpa) gene or its putative receptor, toll-like receptor 2 (TLR2), manifested a delay (~12 h) in parturition associated with decreased induction of contractile gene expression in myometrium.
In light of our findings that steroid receptor coactivators (SRC) are important for transcriptional upregulation of SFTPA in fetal lung type II cells (Fig. 1) and those of O’Malley and colleagues that Src-1/Src-2 double knockout (dKO) mice die at birth of respiratory distress, we crossed mice that were doubly heterozygous for Src-1 and Src-2 (Src-1/-2 dhet). Intriguingly, these mice manifested a striking delay in parturition (~38 h), which was far more pronounced than that of Sftpa-deficient mice (~12 h). The parturition delay in Src-1/-2 double-deficient (Src-1/-2d/d) mice was associated with a marked decrease in SP-A levels in fetal lungs and AF and reduced levels of NF-κB activation, CAP gene expression and PGF2α levels in maternal myometrium. Circulating maternal P4 also was elevated at term, due to a failure of luteolysis. Remarkably, wild-type (WT) females bred to Src-1/-2d/d males manifested similarly delayed labor with decreased activation of NF-κB and CAP genes and elevated P4, strongly suggesting that the defect responsible for the delay in labor is fetal in origin.
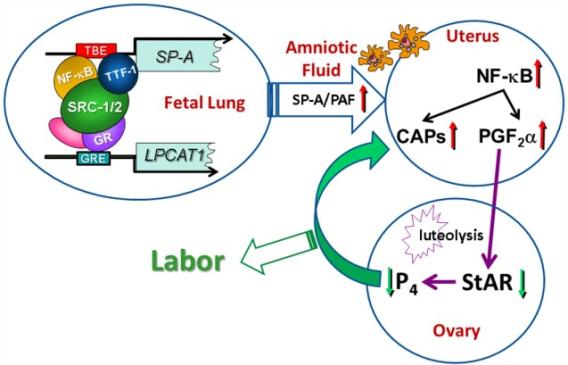 Fig. 6: SRC-1/2 regulation of SP-A and LPCAT1 in fetal lung promote a signaling cascade leading to the initiation of labor.
Fig. 6: SRC-1/2 regulation of SP-A and LPCAT1 in fetal lung promote a signaling cascade leading to the initiation of labor. The pronounced delay in labor (~38 h) in mice carrying Src-1/-2d/d fetuses, compared to that of Sftpa-deficient mice (~12 h), suggests that signaling molecules in addition to SP-A were altered by Src-1/-2d/d. Importantly, levels of the inflammatory phospholipid, platelet-activating factor (PAF), previously implicated in the initiation of parturition, were significantly decreased in the fetal lungs and AF of Src-1/-2d/d fetuses compared to WT. As noted, Src-1/-2 dKO mice die at birth of alveolar collapse/atelectasis, suggesting that surfactant glycerophospholipids also may be altered. In this regard, we found that AF levels of the major surfactant glycerophospholipid, dipalmitoylphosphatidylcholine (DPPC), were significantly reduced in Src-1/-2 dKO fetuses compared to WT. Furthermore, the glucocorticoid-regulated enzyme, lysophosphatidylcholine acyltransferase 1 (LPCAT1), which catalyzes synthesis of both DPPC and PAF, also was significantly decreased in lungs of Src-1/-2d/d fetuses vs. WT.
Based on our collective findings, we propose that towards term, there is increased activation and binding of key transcription factors (e.g. TTF-1 and NF-κB) to response elements upstream of the SFTPA gene and of glucocorticoid receptor (GR) to response elements upstream of the LPCAT1 gene (Fig. 6). These transcription factors recruit SRC-1 and SRC-2, resulting in increased SP-A and LPCAT1 expression and secretion of SP-A and PAF into AF. These immune modulators activate immune cells that migrate to the maternal uterus leading to an activation of inflammatory transcription factors (e.g. NF-κB and AP-1). This contributes to the decline in PR function, with increased CAP gene expression and synthesis of PGF2α, which circulates to the ovary to inhibit steroidogenic acute regulatory protein (StAR), promote luteolysis and cause a decline in circulating P4. The decrease in P4/PR function causes further upregulation of CAP gene expression culminating in labor (Fig. 6).
Currently, we are testing the hypothesis that upon secretion by fetal lung, SP-A and PAF bind to and alter the polarization state of MΦ, which also arise from the fetal lung. We further propose that these MΦ traffic to the maternal decidua, where they alter the decidual leukocyte landscape and trigger a cascade of inflammatory signals leading to a break in maternal-fetal tolerance culminating in parturition.