One of our main focuses is generation of new pluripotent stem cells (PSCs) for novel insights in basic biology and developing new translational applications.
Pluripotency—the ability to give rise to all cell lineages of an adult organism—can be recapitulated in vitro by a spectrum of pluripotent stem cell states stabilized with different culture conditions. Their distinct spatiotemporal characteristics provide an unprecedented tool towards the study of early mammalian development. By modulating the culture parameters and chemical screening, we have been able to generate several novel PSC types that show distinct molecular and functional properties [1-3].
We plan to continue exploring the effects of growth factors, cytokines, extracellular substrates, and physicochemical nature of the in vitro stem cell niche, on the molecular and functional properties of cultured PSCs. This work will enable us to create novel stable PSCs from early embryos with different timing and/or topological properties, which will provide invaluable in vitro models to study the regulatory programs that underlie ontogenesis in vivo.
Most mammalians species do not produce PSCs capable of forming stable chimeras, with the exception of a few rodents and primates. This scarcity severely hampers our understanding of mammalian pluripotency and development.
Recently, we efficiently derived stable embryonic stem cells from bovine blastocysts. This capability opens new avenues for various agricultural and biotechnological applications, e.g. the generation of genetically superior livestock [4].
We seek to develop more culture conditions for deriving PSCs from a variety of extant, endangered, and extinct mammalian species, using in vitro differentiation and/or in vivo interspecies chimera formation. We can then use these PSCs for comparative molecular and functional analyses of early developmental programs in different species.
References
- Kim H, Wu J, Ye S, Tai C-I, Zhou X, Yan H, et al. (2013) Modulation of β-catenin function maintains mouse epiblast stem cell and human embryonic stem cell self-renewal. Nature Communications. 4: 2403.
- Wu J, Okamura D, Li M, Suzuki K, Luo C, Ma L, et al. (2015) An alternative pluripotent state confers interspecies chimaeric competency. Nature. 521: 316–321.
- Yang Y, Liu B, Xu J, Wang J, Wu J, Shi C, et al. (2017) Derivation of Pluripotent Stem Cells with In Vivo Embryonic and Extraembryonic Potency. Cell. 169: 243–257.
- Bogliotti YS, Wu, J, Vilarino M, Okamura D, Soto DA, Zhong C, Sakurai M, Sampaio RV, Suzuki K, Izpisua Belmonte, JC, Ross, PJ. (2018) Efficient derivation of stable primed pluripotent embryonic stem cells from bovine blastocysts. Proc. Natl. Acad. Sci. (In press)
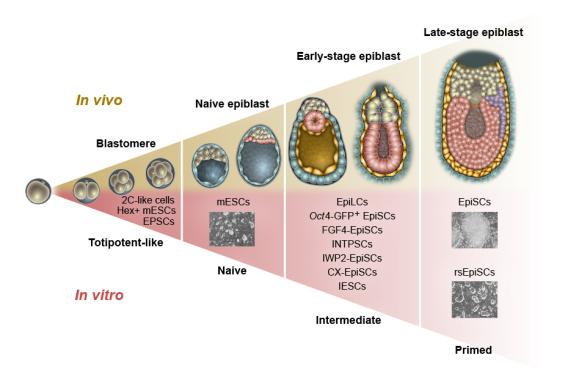 Spatiotemporally distinct pluripotent stem cells: Pluripotent stem cells showing different timing and spatial properties have been isolated from the early mouse embryos
Spatiotemporally distinct pluripotent stem cells: Pluripotent stem cells showing different timing and spatial properties have been isolated from the early mouse embryos