We seek a holistic understanding of adaptive immunity by studying lymphocyte homeostasis at the systemic, cellular, and molecular levels. This work combines genetic animal and translational models with biochemical, biophysical, and immunological methods. Our goal is to better understand immunomodulation in cancer and autoimmunity.
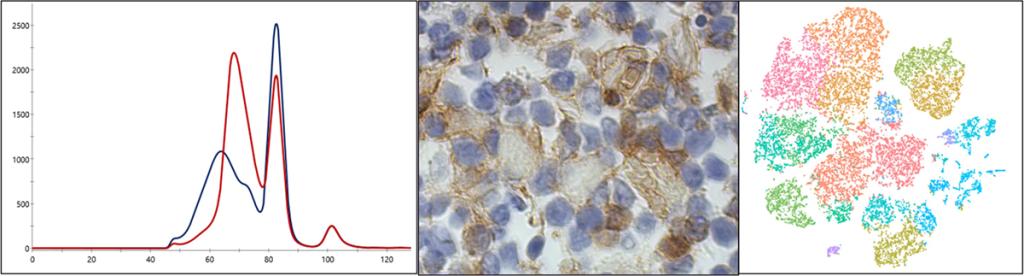
Control of lymphocyte quiescence and survival by Pacs1-Wdr37
We previously discovered that a protein complex containing Pacs1-Wdr37 is critical for normal subcellular calcium handling in lymphocytes. Disruption of this complex impaired lymphocyte development and survival of mature cells but minimally affected lymphocyte activation. Current work in the lab combines subcellular calcium measurements with mouse genetic models to discover how precise regulation of calcium handling influences the persistence of normal, autoreactive, and malignant lymphocytes.
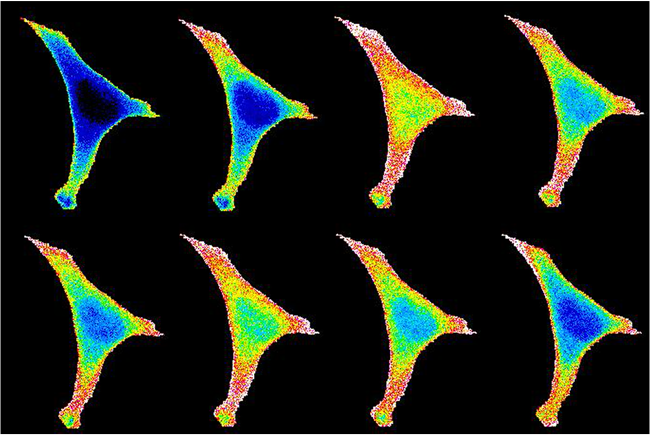
Regulation of CD8 T cell fate by orphan cell surface receptors
Lymphocyte activation leads to the differentiation of memory cells characterized by their long lifespan, self-renewal capabilities, and rapid recall response. We are investigating how extracellular signals through orphan receptors on CD8 T cells can limit their differentiation and survival. Current work in the lab combines cellular and molecular immunological methods with proteomic and transcriptomic analyses to determine how novel inputs into lymphocyte signaling networks can modulate cell fate.
Join Our Lab
If you have an interest in the research that we are conducting or if you would like to be considered for a position in the lab, please send an email.