Peritrichous bacteria such as E. coli and Salmonella species historically have served as models for understanding many aspects of flagellar gene transcription and biosynthesis. Alternatively, some important bacterial pathogens such as Campylobacter jejuni, Helicobacter pylori, Vibrio cholerae, and Pseudomonas aeruginosa are polar flagellates. These bacteria are genetically programmed to produce only a limited number of flagella in polar regions to create specific flagellation patterns. V. cholerae and P. aeruginosa synthesize a single flagellum only at one pole (monoflagellation), C. jejuni produces a single flagellum at both poles (amphitrichous flagellation), and H. pylori produces a tuft of flagella at a single pole (lophotrichous flagellation).
Polar flagellates possess a different repertoire of regulatory factors than peritrichous bacteria to accurately control the transcription of flagellar genes, whose regulation is linked to specific steps in flagellar biogenesis. Both proper transcriptional regulation of flagellar genes and production of the correct flagellation pattern are required by polar flagellates for swimming motility to thrive in the environment and infect hosts.
We have used Campylobacter jejuni as a model system to elucidate a general mechanism present in many polar flagellates that coordinates transcription of genes for polar flagella in relation to steps in polar flagellar biogenesis. We discovered a conserved sensing mechanism in similar two-component signal transduction systems (TCSs) in polarly-flagellated pathogens that include C. jejuni and Helicobacter pylori FlgSR TCS, Vibrio cholerae FlrBC TCS, and P. aeruginosa FleSR. We found that the sensor histidine kinase of these systems detects the formation of the flagellar type III secretion system (T3SS), MS ring, and rotor as an early regulatory checkpoint in polar flagellar biogenesis. Once this structure is formed, the TCSs sense this structure to then activate sigma54-dependent flagellar rod and hook expression for polar flagellar biogenesis to proceed.
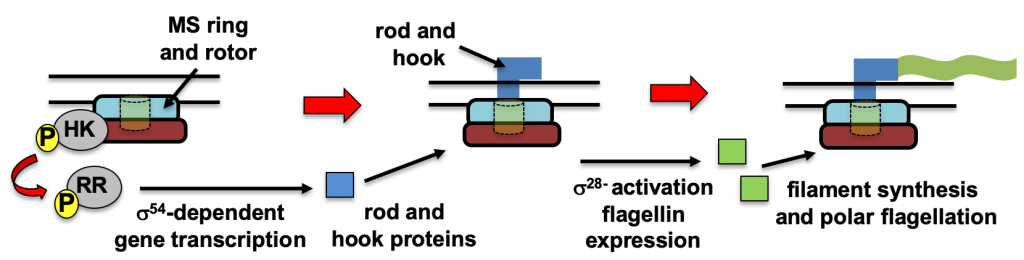 Signal transduction pathway mediated by the conserved FlgSR, FlrBC, and FleSR two-component systems of polar flagellates for polar flagellar synthesis.
Signal transduction pathway mediated by the conserved FlgSR, FlrBC, and FleSR two-component systems of polar flagellates for polar flagellar synthesis. We are currently exploring how numerical and spatial regulation of flagellar biogenesis occurs in polarly-flagellated bacterial pathogens. To maintain amphitrichous flagellation in C. jejuni, a single flagellum must be synthesized at the new pole of daughter cells after division. Among the factors we are investigating for spatial and numerical control of polar flagellar biogenesis in C. jejuni are the FlhF GTPase and the FlhG ATPase, which are common to other polar flagellates. Furthermore, we discovered that FlhG and flagellar substructures also impact the ability of C. jejuni to divide properly. This finding indicates that the flagellum influences spatial regulation of the divisome, which is required for the proper symmetrical division to generate viable daughter cells. We expect our work to reveal new mechanisms by which bacteria organize their cell bodies and mediate spatial and numerical regulation of macromolecular complexes that are essential for many biological activities.
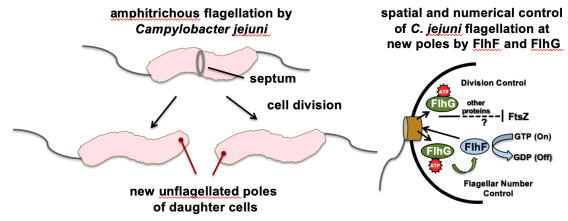 Amphitrichous flagellation at new poles of C. jejuni cells after division.
Amphitrichous flagellation at new poles of C. jejuni cells after division.  Cell division and flagellar number defects in C. jejuni mutants.
Cell division and flagellar number defects in C. jejuni mutants. Relevant Publications
Transcription of s54-dependent but not s28-dependent flagellar genes in Campylobacter jejuni is associated with formation of the flagellar secretory apparatus
Hendrixson, D. R., and DiRita, V. J. Mol Microbiol (2003) 50:687-702
Restoration of flagellar biosynthesis by varied mutational events in Campylobacter jejuni
Hendrixson, D. R. Mol Microbiol (2008) 70:519-536
Activation of the FlgSR two-component system of Campylobacter jejuni is linked to the flagellar export apparatus
Joslin, S. N., and Hendrixson, D. R. J. Bacteriol (2009) 191:2656-2667
FlhF and its GTPase activity are required for distinct processes in flagellar gene regulation and biosynthesis in Campylobacter jejuni
Balaban, M., Joslin, S. N., and Hendrixson, D. R. J. Bacteriol (2009) 191:6602-6611
Polar flagellar biosynthesis and a regulator of flagellar number influence spatial parameters of cell division in Campylobacter jejuni
Balaban, M. and Hendrixson, D. R. PLoS Pathog (2011) 7:e1002420
A specificity determinant for phosphorylation in a response regulator prevents in vivo crosstalk and modification by acetyl phosphate
Boll, J. M., and Hendrixson, D. R. Proc Natl Acad Sci USA 9(2011) 108:20160-20165
A regulatory checkpoint during flagellar biogenesis in Campylobacter jejuni initiates signal transduction to activate transcription of flagellar genes
Boll, J. M., and Hendrixson, D. R. mBio (2013) 4:e00432-13
FlhG employs diverse intrinsic domains and influences FlhF GTPase activity to numerically regulate polar flagellar biogenesis in Campylobacter jejuni
Gulbronson, C. J., Ribardo, D. A., Balaban, M. Knauer, C., Bange, G., and Hendrixson D. R.
Mol Microbiol (2016) 99:291-306
A polar flagellar transcriptional program mediated by diverse two-component signal transduction systems and basal flagellar proteins is broadly conserved in polar flagellates
Burnham, P. M., Kolar, W. P., and Hendrixson, D. R. mBio (2020) 11:e03107-19